Platform Technology

Overview
We are engaging our Thin Film Freezing technology to transform medicines into elegant and potent dry powders for better efficacy, safety, and stability.
The Challenge
A therapy is only as effective as its ability to reach its target. Many drugs do not adequately reach their target due to inferior physical properties, leading to compromised bioavailability, slow onset of action, and suboptimal efficacy. Additionally, poorly water-soluble drugs are often given in high doses, leading to high risk of toxicities and adverse side effects.
Nearly 40% of marketed drugs and 90% of drugs in the discovery pipeline are poorly water-soluble, which makes these drugs difficult to formulate. Many biologic-based therapies, including proteins, RNA, vaccines, viruses, and others, face stability, structure, and activity problems when delivered in liquid form.


Our Solution
Our proprietary technology, Thin Film Freezing, was designed to generate dry powders that overcome these challenges. This technology can enable therapeutic delivery directly to the target organ, such as the lungs, which can improve, for example, the absorption of poorly water-soluble drugs and enhance the pharmacokinetic effects and safety profile of these therapies. Our Thin Film Freezing technology can improve the delivery of small molecules, biologics, and combination therapies.
Our technology enables the formation of “Brittle Matrix Particles” that possess low bulk density, high surface area, and amorphous or nanocrystalline morphology. These attributes ensure that a therapy maintains its structure, activity, and functionality, as well as its thermostability and aerosol properties for delivery to different parts of the body, including the deep lung. The aerodynamic properties of these particles can enable the portion of drug deposited to the target organ to reach as high as 75%.
Thin Film Freezing creates engineered particles that can provide site-specific dosing. For example, these particles may be used for needle-free administration of vaccines or to lower the total doses of drugs previously given orally to reduce unwanted side effects.
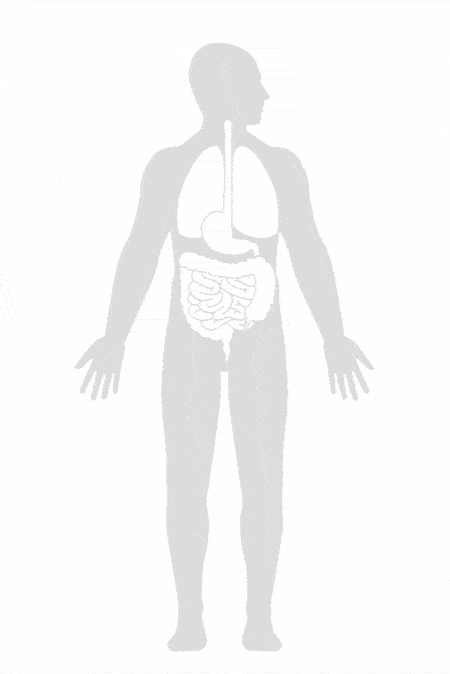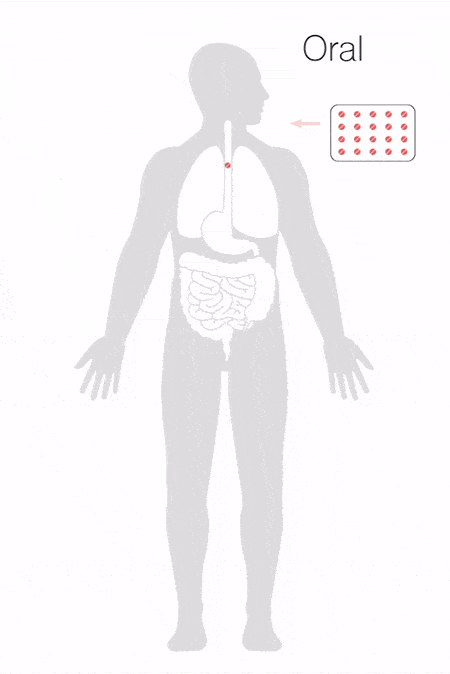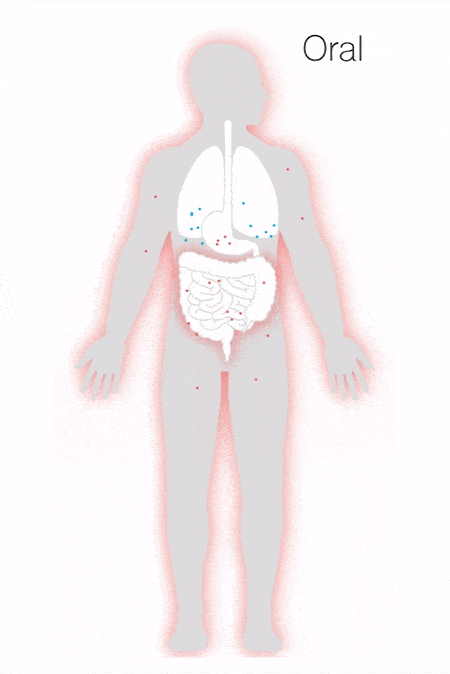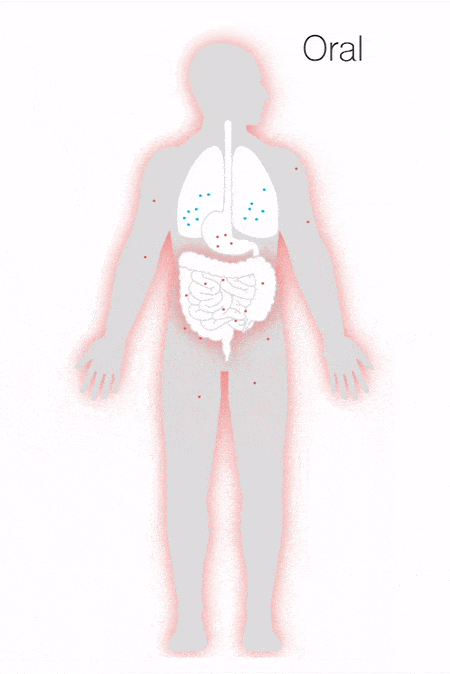


How Thin Film Freezing Works


1. Formulation: The Thin Film Freezing process involves first combining a drug or drugs in a liquid carrier system, which can include agents designed to promote dispersion and avoid aggregation, as well as excipients to promote delivery to the target site.


2. Freezing: Controlled freezing the liquid material on the surface of a cryogenic drum results in Brittle Matrix Particles: highly porous, large surface area, low-density particles.


3. Drying: The frozen solvent is then removed by sublimation to yield a dry powder material that can be aerosolized to facilitate delivery to the lungs, nose, eyes and topically through the skin.


4. Finishing: The Brittle Matrix Particles are then further processed to facilitate creation of the final dosage form for the targeted route of administration.
TFF products demonstrate a clear advantage over other dry powder treatments. The process may use industry standard solvents, lung-approved excipients, and conventional processing equipment in addition to TFF’s custom-made cryogenic drum. Every formulation and program is unique.
See our platform in action:

Benefits
Efficacy
Thin Film Freezing transforms therapies into elegant dry powder formulations that can be delivered directly to the site of need, often circumventing systemic administration and leading to improved bioavailability, faster onset of action, and improved safety and efficacy.
Safety
Thin Film Freezing converts therapies into a dry powder formulation that may contain a high level of active drug, unlocking the potential to administer a lower effective dose with an improved safety profile.
Broad applicability
Thin Film Freezing technology has universal applicability to large and small molecules, vaccines, and biologics. We are transforming medicines into stable dry powders that may be reconstituted for injection or enable alternatives to injection, including intranasal, inhalation, intraocular and topical delivery.
Many of our Thin Film Freezing formulations unlock the opportunity to administer treatment more easily at home without the assistance of a healthcare professional, for example through delivery of treatment via a pocket-sized dry powder inhaler.
Stability
Thin Film Freezing converts vaccines and other biological products that are traditionally delivered in liquid form into a dry powder, which is more stable at room temperature. This eliminates the need for ultra-cold chain refrigeration, a major logistical challenge to distribution and global accessibility.
We can apply our Thin Film Freezing technology to improve the stability of finished biological dosage forms and overcome the limitations of current biologic manufacturing, facilitating rapid scale-up and rollout of new products.